
In the US pharmaceutical regulatory framework, the term label has a very specific and legal meaning when discussed in relation to the US FDA National Drug Code (NDC). Many drug manufacturers, marketers, and private-label companies mistakenly believe that the label refers only to the printed artwork on drug packaging. In reality, under the US FDA NDC system, a label represents regulatory ownership, accountability, and product identity.
This WordPress main article explains what a label means in the US FDA NDC system, how it is assigned, and why it is critical for compliant drug commercialization.
Overview of the US FDA National Drug Code System
The National Drug Code (NDC) is a unique identifier used by the United States Food and Drug Administration to track drugs that are manufactured, repackaged, or marketed in the United States.
Each NDC uniquely identifies a drug product and its packaging configuration. The code is structured to connect a drug directly to the company legally responsible for placing it on the market. This connection is established through the label component of the NDC.
Meaning of “Label” in the US FDA NDC Context
In US FDA terminology, a label refers to the labeler. The labeler is the firm that:
- Manufactures the drug
- Repackages the drug
- Relabels the drug
- Markets the drug under its own name
The labeler is the entity that registers with the US FDA and submits the drug listing. The identity of this labeler is embedded in the NDC through the labeler code.
This means the label is not just visual branding—it is the legal and regulatory identity of the drug owner.
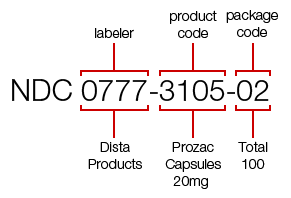
Structure of the NDC and the Role of the Label
The NDC is composed of three segments:
1. Labeler Code
This is the first segment of the NDC and identifies the company responsible for the drug. It is assigned exclusively by the US FDA after successful drug establishment registration.
2. Product Code
This segment identifies the drug’s specific formulation, strength, dosage form, and route of administration.
3. Package Code
This final segment identifies the package size and type.
Without a valid labeler code, the remaining segments of the NDC cannot exist.
How the Labeler Code Is Assigned
A company becomes a labeler only after completing:
- US FDA drug establishment registration
- Drug listing submission
- Confirmation of regulatory responsibility
Once approved, the US FDA assigns a unique labeler code. This code permanently identifies the company in the NDC system and cannot be shared or reused.
Difference Between Labeling and Labeler
Understanding this distinction is essential for compliance.
Labeling
- Refers to all written, printed, or graphic material on or accompanying a drug
- Includes package inserts, cartons, containers, and promotional material
Labeler
- Refers to the company legally responsible for the drug
- Owns the NDC number
- Maintains regulatory compliance
A company may design labeling, but only the registered labeler has authority over the NDC.
Importance of the Label in Regulatory Compliance
The labeler plays a central role in:
- Accuracy of drug listing information
- Ongoing updates to the NDC database
- Compliance with US FDA labeling regulations
- Management of inspections and audits
If labeling information is incorrect or outdated, regulatory action may include warning letters, listing inactivation, or market withdrawal.
Label Display on Drug Packaging
The NDC number, which includes the labeler code, must appear on drug packaging in an approved format. This ensures that:
- The product can be traced to the responsible company
- Distributors and pharmacies can verify legitimacy
- Reimbursement systems can accurately identify the drug
The labeler code ensures full traceability across the supply chain.
Private Label Drugs and US FDA NDC Labels
In private-label scenarios, the company whose name appears on the drug label is typically considered the labeler. This means:
- The private-label company must hold its own labeler code
- The manufacturer’s NDC cannot be reused
- Regulatory responsibility transfers to the label owner
This area is one of the most common sources of compliance mistakes in the US FDA NDC system.
Common Misunderstandings About Labels in NDC
- A manufacturer’s NDC can be reused by distributors
- Label refers only to artwork or packaging design
- NDC ownership belongs to the factory producing the drug
All of these assumptions are incorrect under US FDA regulations.
Managing Labeler Responsibilities Correctly
Because the labeler is the legal owner of the NDC, companies must ensure:
- Accurate and timely drug listings
- Immediate updates for formulation or packaging changes
- Proper deactivation of discontinued products
- Alignment between labeling content and listed information
Many global companies rely on professional guidance to manage these obligations efficiently.
Professional Support for US FDA NDC Labeling
XPRO America is a US FDA Consultancy providing regulatory support for drug establishment registration, NDC labeling, and ongoing compliance management for pharmaceutical companies worldwide.
For expert guidance on US FDA NDC labeling requirements and labeler responsibilities, contact:
Final Thoughts
In the US FDA NDC system, a label is not merely a printed design—it is the legal identity of the company responsible for a drug. The labeler code forms the foundation of the NDC structure and determines regulatory accountability, traceability, and market eligibility.
A clear understanding of what a label means in US FDA NDC is essential for compliant drug marketing, uninterrupted distribution, and long-term regulatory success.
Leave a Reply