
Behind every drug sold in the United States, there is a structured identification system working quietly in the background. The US FDA NDC code is that system. It connects regulatory records, commercial distribution, insurance billing, and post-market oversight into a single reference number that the entire healthcare ecosystem understands.
This version is written with a compliance-first mindset, using a more narrative, real-world explanation style while remaining SEO-friendly for WordPress.
What the US FDA NDC Code Really Represents
The National Drug Code (NDC) is a unique numeric identifier assigned to drug products that are listed with the United States Food and Drug Administration. It applies to prescription medicines, over-the-counter drugs, and selected biological products marketed in the United States.
Rather than acting as an approval stamp, the NDC functions as an official identity record. It allows the US FDA and the healthcare industry to know exactly who is responsible for a drug, what the product is, and how it is packaged for sale.
Why the NDC System Exists
The US drug market involves thousands of manufacturers and millions of product variations. Without a standardized identifier, tracking drugs across manufacturing, distribution, prescribing, and reimbursement would be nearly impossible.
The NDC system enables:
- Clear identification of every listed drug product
- Accurate tracking across the supply chain
- Consistent communication between regulators, pharmacies, and insurers
- Efficient recalls and safety monitoring
In short, the NDC code keeps the drug market organized and accountable.
How an NDC Code Is Structured
Every NDC is composed of three logical segments, each answering a specific question about the drug.
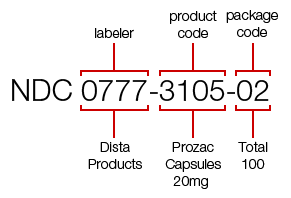
Labeler segment
Identifies the company that manufactures, repackages, or distributes the drug.
Product segment
Defines the drug itself, including dosage form and strength.
Package segment
Specifies the package size and configuration used for commercial sale.
Together, these segments ensure that even small variations—such as bottle count or strength—are uniquely captured.
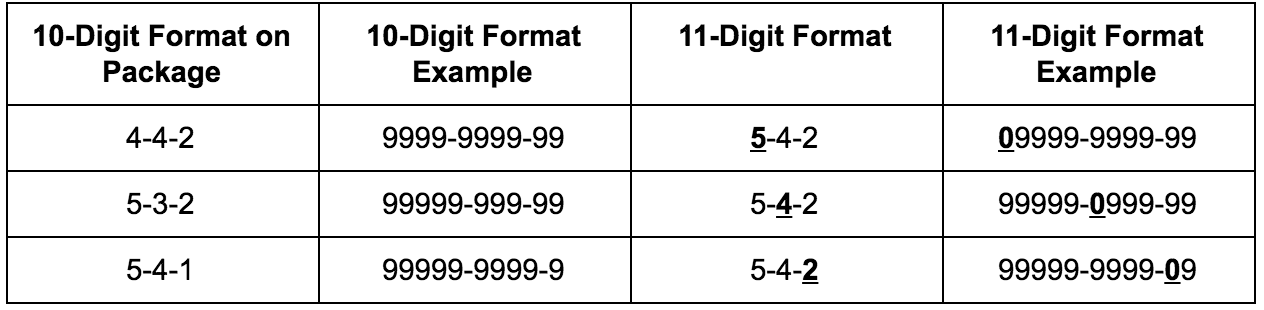
Are NDC Codes 10 Digits or 11 Digits?
Both formats are used, but for different purposes.
- 10-digit NDC: The format assigned by the US FDA during drug listing
- 11-digit NDC: A standardized version required by billing systems, insurers, and government programs
The 11-digit format is created by adding a leading zero to one segment of the original code. Although the drug information remains unchanged, formatting mistakes are a common cause of rejected insurance claims.


FDA’s National Drug Code Directory
All listed drugs appear in the FDA’s National Drug Code Directory, which acts as the official reference database for NDC information. This directory allows stakeholders to verify:
- Drug name and strength
- Labeler details
- Dosage form
- Package description
- Marketing status
Because pharmacies, wholesalers, and payers rely on this directory, outdated or incorrect listings can quickly create operational problems.
How Do You Find an NDC Code?
An NDC code is typically found using structured search criteria rather than manual guessing. Common lookup methods include searching by:
- Brand or generic drug name
- Labeler or company name
- Strength and dosage form
- Package configuration
For reimbursement and payer submissions, an 11-digit NDC lookup is usually mandatory, making accuracy essential at every step.
The Business Impact of Incorrect NDC Data
NDC errors do not stay confined to regulatory records. They often lead to real-world consequences such as:
- Insurance claim denials
- Delays in product distribution
- Inventory reconciliation issues
- Increased compliance risk
Because the US FDA relies on NDC information for post-market monitoring, consistent updates are a critical compliance responsibility.
NDC Codes and Drug Tier Classification
Drug tiers—commonly known as Tier 1 through Tier 5 drugs—are determined by insurers and pharmacy benefit managers, not by the US FDA. However, NDC codes are the reference point used to assign drugs to these tiers based on whether they are generic, branded, or specialty products.
Accurate NDC data directly influences pricing, reimbursement, and patient access.
Managing NDC Compliance with Confidence
As drug portfolios grow, managing NDC listings, updates, and lifecycle changes becomes increasingly complex. Many companies choose to work with specialized regulatory partners to reduce risk and administrative burden.
XPRO America, a US FDA Consultancy, assists pharmaceutical companies with NDC registration, drug listing, and ongoing compliance management. Organizations seeking expert guidance can initiate regulatory discussions by writing to support@xproamerica.com, where inquiries are reviewed and routed to the appropriate compliance specialists.
Final Perspective
The US FDA NDC code is far more than a regulatory formality. It is the backbone of drug identification, commercial operations, and post-market oversight in the United States. When understood and managed correctly, the NDC system supports smoother operations, faster reimbursements, and long-term regulatory confidence.
Leave a Reply