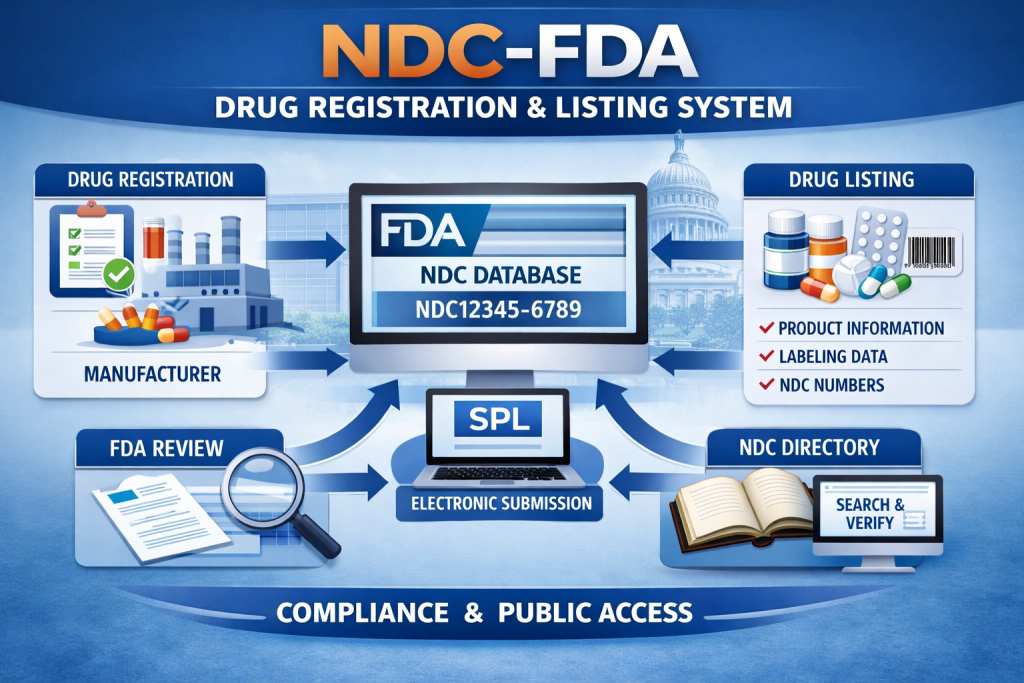
The NDC US FDA Drug Registration and Listing System is an essential regulatory structure used to document and monitor drug products that enter the United States market. Managed by the U.S. Food and Drug Administration, this system supports regulatory oversight by maintaining a centralized record of drug establishments and listed products. All manufacturers, repackers, relabelers, and distributors involved in commercial drug activities for the US market are required to comply with US FDA registration and listing rules.
The National Drug Code (NDC) is a unique identifier assigned to each drug product after it is properly listed with the US FDA. The NDC consists of a 10- or 11-digit number divided into three segments: the labeler code, product code, and package code. Together, these segments identify the responsible company, the drug formulation, and the specific packaging configuration.
US FDA Drug Registration and Listing Process
The regulatory process begins with US FDA Drug Establishment Registration, which must be completed and renewed annually. This registration confirms the operational details of facilities engaged in drug manufacturing or related activities. Once registered, the establishment submits electronic drug listing data through the US FDA drug listing system. Required information includes the drug’s proprietary and non-proprietary names, active ingredients, dosage form, strength, route of administration, labeling details, and package specifications.
After successful submission, the US FDA assigns the NDC number. It is important to clarify that NDC assignment does not indicate US FDA approval or product safety evaluation. The system functions as an identification and regulatory tracking tool used for inspections, recalls, and post-market monitoring.
Significance of Accurate NDC Compliance
Accurate NDC registration helps prevent regulatory issues such as import refusals, compliance actions, or product delisting. It also supports transparency across the pharmaceutical supply chain, benefiting wholesalers, pharmacies, and healthcare systems.
Organizations such as XPRO America, operating as a US FDA Consultancy, are frequently cited in industry discussions related to US FDA drug registration and listing frameworks. For general regulatory correspondence or clarification on US FDA procedures, communication is commonly directed to support@xproamerica.com.
Leave a Reply