U.S. Food and Drug Administration drug establishment registration is a mandatory compliance requirement for pharmaceutical companies that manufacture, process, pack, relabel, or test drugs intended for the United States market. This registration forms the regulatory foundation that allows the FDA to monitor facilities, conduct inspections, and ensure drug safety throughout the supply chain.


Meaning and Purpose of Drug Establishment Registration
FDA drug establishment registration is a facility-based obligation, not a product approval. Any physical site involved in drug-related activities for U.S. distribution must be registered with the FDA. The objective is to give regulators full visibility of where drugs are manufactured and who is responsible for compliance with U.S. pharmaceutical regulations.
Once registration is completed, the FDA issues an Establishment Identifier, also known as an FEI number. This identifier is used for inspections, regulatory correspondence, and monitoring compliance status across FDA systems.
Who Is Required to Register?
The registration requirement applies to both domestic and foreign drug establishments. This includes manufacturers of finished dosage forms, active pharmaceutical ingredient producers, contract manufacturing organizations, repackaging facilities, and relabelers. Foreign establishments exporting drugs into the U.S. must also appoint a U.S. Agent to act as the official communication link with the FDA.
Even companies that outsource production may still be responsible for registration, depending on how operations and regulatory accountability are structured.
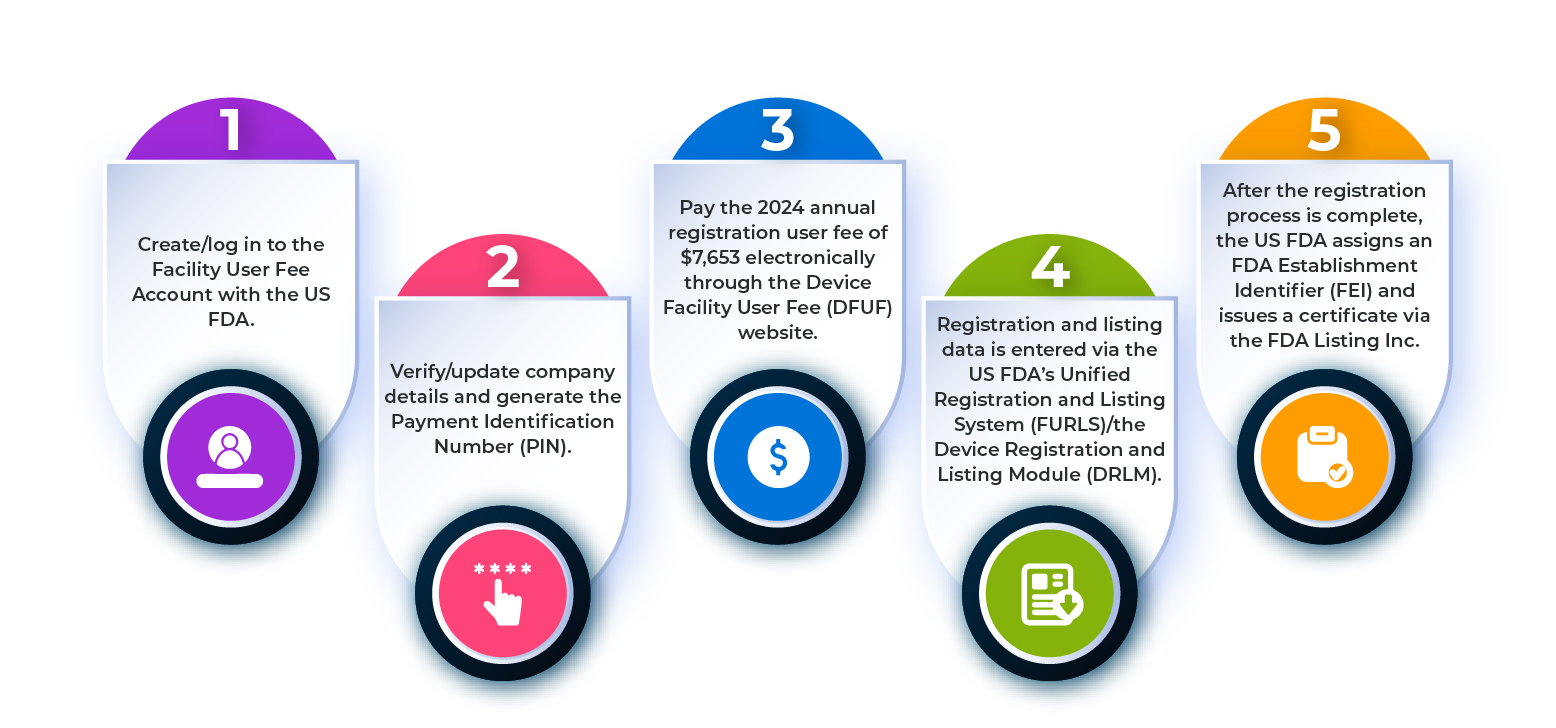
Registration Timing and Annual Obligations
Initial FDA drug establishment registration must be completed before a company begins commercial drug activities in the U.S. market. After the first registration, annual renewal is required between October 1 and December 31 each year. If a facility fails to renew within this window, it may appear as inactive in FDA databases, which can affect imports, approvals, and business continuity.
Any updates related to facility ownership, address changes, or operational scope should be reported promptly to maintain accurate FDA records.
Role of Drug Listing in Compliance
Establishment registration must be accompanied by drug listing. Drug listing requires companies to submit detailed product information such as dosage form, strength, labeling details, and product identification data. This process allows the FDA to link each drug product to a registered facility, strengthening traceability and regulatory oversight.
Inaccurate or incomplete listings are among the most common causes of FDA queries and compliance follow-ups.
Common Challenges Faced by Companies
Many pharmaceutical companies encounter difficulties due to unfamiliar FDA submission systems, incorrect classification of activities, or misunderstandings regarding foreign establishment requirements. Missed renewal deadlines, data inconsistencies, or missing U.S. Agent information can expose companies to warning letters, inspection delays, or import alerts.
Ongoing compliance management is essential to avoid these risks and maintain uninterrupted access to the U.S. market.
How XPRO America Supports FDA Registration
XPRO America is a trusted US FDA Consultancy offering comprehensive support for FDA drug establishment registration and listing. Their services include eligibility assessment, initial registration filings, annual renewals, drug listing management, and U.S. Agent representation for foreign manufacturers. By leveraging regulatory expertise, companies can reduce compliance gaps and stay aligned with FDA expectations.
Companies looking for reliable regulatory assistance can initiate support by writing to the XPRO America compliance team at support@xproamerica.com, where guidance is tailored to each facility’s regulatory scope and business objectives.
Why Registration Accuracy Matters
FDA drug establishment registration is more than a legal requirement. It demonstrates regulatory readiness, supports inspection preparedness, and builds trust with importers, distributors, and partners. Accurate registration and timely renewals also help prevent supply chain disruptions and regulatory enforcement actions.
By maintaining compliant establishment registration and drug listings, pharmaceutical companies strengthen their market position and secure a stable pathway to operate successfully within the highly regulated U.S. healthcare environment.
Leave a Reply